Belfast-based medical technology company Neurovalens has achieved EU Medical Device Regulation (MDR) compliance and UK Medicines and Healthcare products Regulatory Agency (MHRA) registration for Modius Sleep, its prescription-only neurostimulation device for treating insomnia, clearing the path for commercial sales across both markets.
The Newsletter reported that Neurovalens is among the first Northern Ireland-based companies to achieve EU MDR certification, placing it alongside some of the world's leading medical device manufacturers in meeting the regulation's stringent safety, clinical evaluation, and quality management standards.
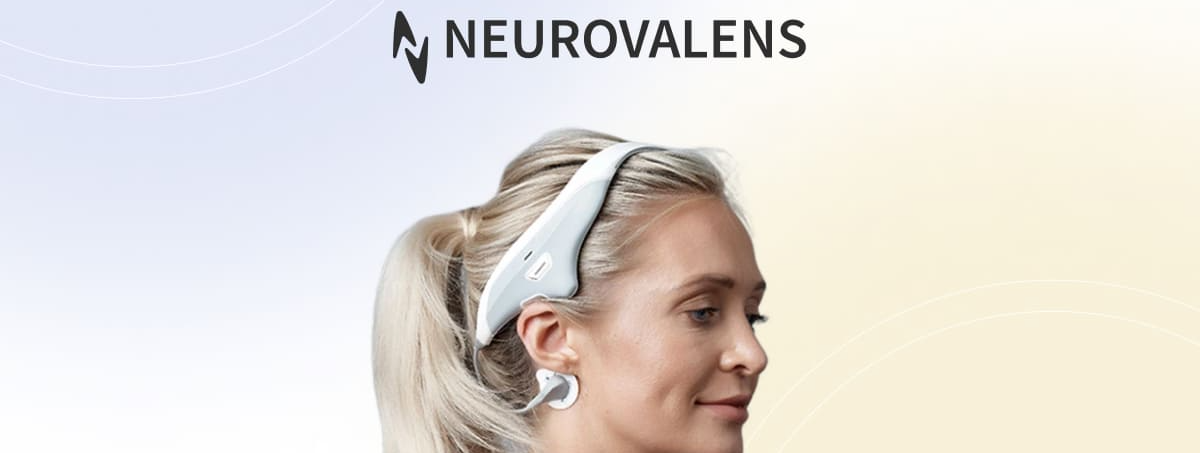
Modius Sleep uses electrical vestibular system stimulation (VSS), delivering a small, safe electrical pulse to the head for 30 minutes before bed to non-invasively stimulate areas of the brain known to influence sleep, allowing users to carry out other activities during treatment.
The device is already available via prescription in the United States for $2,265 (€1,957.75), where it has been in use since receiving FDA approval in 2023, and is also available to US veterans through the Department of Veterans Affairs.
Neurovalens has raised approximately £20 million in equity and debt funding to date from investors including Wharton Asset Management, IQ Capital, Techstart Ventures, and the British Business Bank, among others.
The company also has regulatory approvals pending for the treatment of post-traumatic stress disorder (PTSD), and continues to run clinical trials on the effectiveness of its technology across further conditions.
Dr Jason McKeown, MD, PhD, CEO of Neurovalens, said: "Most other sleep-related products on the market measure the symptoms of insomnia, but Modius Sleep is a non-invasive device that actually treats the underlying issue, which can have a transformative impact on the lives of our patients."
Get the full details of Neurovalens' EU and UK regulatory approval for Modius Sleep.

















.png)
